The Côte d’ivoire Ethics Capacity Building Initiative (CECaBI) is a consortium of different partners whose objectives are to strengthen the knowledge and skills of the senior members of the National Ethics Committee for Life Sciences and Ivorian Authorities of Pharmaceutical Regulation (Autorités Ivoiriennes de Régulation Pharmaceutique (AIRP)), on how to conduct high-quality ethics reviews, ensure optimal protection of research participants, and effectively manage and coordinate clinical trials.
Pharmalys is part of this initiative. As such, Karim Bagaté, Director of Clinical Operations & Safety delivered a capacity building programme in Côte d’Ivoire in 2023. The training was delivered in two parts. The first one, in March 2023, was about the Good Clinical Practice (GCP) guidelines and site inspection. The second part, in October 2023 was a refresher on GCP with very practical exercises and workshops and a site inspection visit.
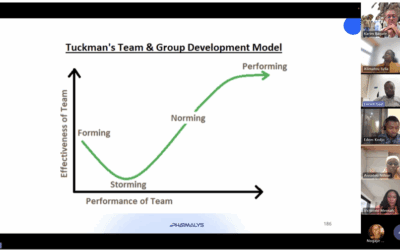
Project Management Training: Pharmalys’ Commitment to Professional Growth
Pharmalys continues to invest in the professional development of its employees through dedicated training initiatives. A recent initiative is the Project Management Training Programme (PMBOK concept), led by Dr. Karim Bagaté, Director of Clinical Operations and Safety...